动物营养方案可保障动物健康及提高生产力
公司新闻 | 2021.03.03

Consumer preferences and government regulations are changing the way you raise livestock and poultry, particularly with regard to the use of antibiotics—no matter where you are located. Yet the need to optimize animal health and efficient growth has never been more important.
The world population has increased to more than 7 billion, 815 million of whom were estimated to be chronically undernourished in 2016.1 As demand for animal protein climbs, so does the need to safely and effectively control the many bacterial diseases that can substantially hamper animal health.
One estimate notes that 80 types of bacteria, such as Escherichia coli, Salmonella and Clostridium, pose serious threats to the poultry industry alone.2 These pathogens are disease-causing culprits in swine, dairy and beef production, too.
Increasingly, due to changing market forces, producers worldwide are focusing on ways to lower their on-farm use of antibiotic tools and searching for non-antibiotic nutritional solutions to tackle animal health challenges and enhance production.
Clostridia Prevalence
To understand appropriate non-antimicrobial responses to animal disease threats, it’s best to gain an understanding of the challenge faced.
In the case of clostridia, these bacteria are normal inhabitants of the soil and poultry and livestock environments across the world—no region or country is exempt. This means animals constantly ingest low levels of clostridia as a result of its widespread, underlying presence. As bacterial loads rise, animals become increasingly vulnerable to high-stress events, creating a tipping point for disease and performance deficiencies.
For example, to date in the United States, more than 69,000 different clostridial isolates, or strains, have been harvested from thousands of dairy fecal and feed samples. These samples were assayed by researchers at ARM & HAMMER.
- About half of these isolates (53.9 percent) make up a well-known toxigenic species, Clostridium perfringens, which can negatively impact gut health and lead to serious digestive issues such as hemorrhagic bowel syndrome (HBS).
- The other isolates (46.1 percent) include Clostridium that produce metabolic end-products that have a negative impact on rumen efficiency. Instances of “off-feed” cows or stomach upsets can often be tied to these secondary effects of clostridia.
The samples were collected from across the U.S. and organized into regions defined by geography and prevailing farm management styles. This created a clear picture of clostridial population diversity in specific regions. Similar collection and testing programs have been adopted for other U.S. animal species, and steps are underway to introduce these processes and solutions to international regions, as well.
Species Diversity
The results indicate that both the species of Clostridium and the level of diversity differ across regions sampled.
Regional differences can be attributed to varying soil types, environments, feedstuffs, animals and management styles. Taken together, these elements form the Microbial Terroir™, or microbial makeup, of the region or an individual farm. Differences in Microbial Terroir help explain why certain challenges plague some areas more than others.
This same principle applies to environments and different animal species everywhere—parallel collection and testing programs for poultry and swine show similar results in bacterial diversity by farm, complex and region. The difference lies in the diseases that afflict the different species—necrotic enteritis in the case of poultry versus HBS in cattle, for example.
What to do?
The aim, then, must be to limit the clostridial load on your farm or complex and change your farm’s pathogen profile to prevent herd or flock health and performance issues.
The robust farm sampling and analysis program undertaken in the United States has also provided a solution for combating clostridia on a regional and individual farm or complex basis.
Utilizing clostridia profiles, researchers have identified several specific combinations of proprietary strains of Bacillusbacteria that inhibit both toxigenic and non-toxigenic Clostridium in different U.S. regions—known as Targeted Microbial Solutions™.
Bacillus have coexisted with clostridia in the soil for eons and are their natural enemies; long-term use results in changes to clostridial population levels and species diversity.
The important lessons are:
- Clostridial populations and diversity vary across the country and across the globe. Different combinations of Bacillus are necessary to address clostridial challenges specific to each region.
- Feeding Bacillus over the long term results in changes to the Microbial Terroir of a farm.
Ultimately, this solution helps change the pathogen profile of a farm or complex, lowers harmful bacterial loads in GI tracts and enhances overall animal health, production and efficiency.
Positively Affect Gut Health
There’s a reason animal agriculture is so focused on gut health.
As Australian researchers note, “The nutritional and health status of poultry are interlinked with gut health which includes immune system, gut microbial balance and macro and micro-structural integrity of the gut. The health of the gastrointestinal tract affects digestion, absorption and metabolism of nutrients, disease resistance and immune response.”3 Researchers of swine, dairy and beef, share similar sentiments.
To aid in the quest for non-antibiotic solutions that help enhance animal health and productivity, producers are increasingly turning to Refined Functional Carbohydrates™ (RFCs™). RFCs have been shown to offer many benefits that may be associated with gut health and immune response.
RFCs are the components harvested from yeast cell walls (S. cerevisiae) using specific enzymes during a proprietary manufacturing process. This enzymatic hydrolysis yields MOS (Mannan Oligosaccharides), Beta 1,3-1,6 glucans and D-Mannose—all of which are readily bioavailable in CELMANAX™ due to this proprietary process. Research4,5,6,7 shows that each RFC has a specific mode of action and outcome when fed to livestock.
Turkey Results
A recent trial8 examined the effects of RFCs on turkey poult performance when they were placed in stressful production environments. During the trial, 204-day-old male turkey poults were distributed in completely randomized replicated pens/treatments.
Treatments included:
- Control (no supplementation)
- RFC (100 g/MT) fed continuously
- RFC (200 g/MT) fed only during first week of placement and for a week encompassing each period of transport stress
- All poults were transported at weeks six, 12 and 16 to mimic industry practice. During weeks of transport, on alternate days, birds were exposed to an environmental challenge of E. coli to emulate field conditions
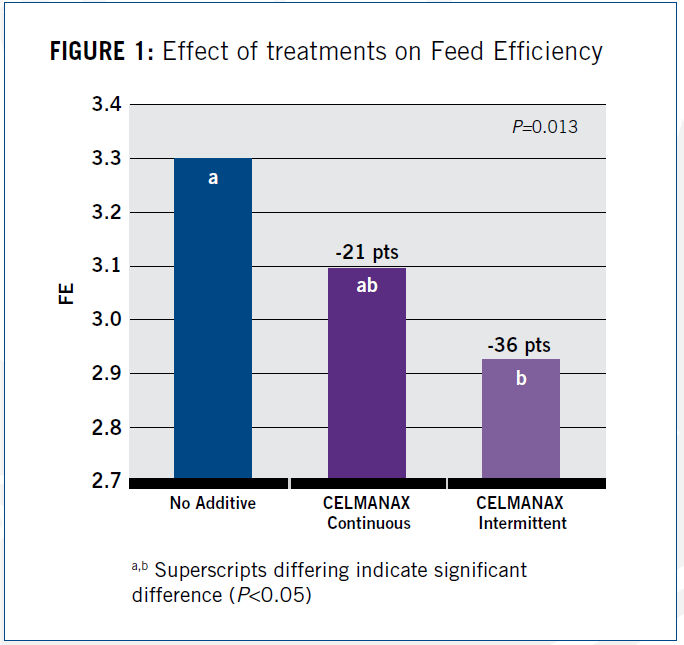
After analyzing the data, researchers found that continuous and intermittent feeding of RFCs improved feed efficiency (FE) by 21 and 36 points, respectively; the intermittent feeding significantly improved FE (P=0.01) as shown in Figure 1.
One point to note. In North America, feed efficiency is calculated by dividing feed intake by weight gain. The lower the number, the more efficient the flock in using the feed supplied. In some other parts of the world, feed efficiency is calculated as weight gain divided by feed intake. In that instance, higher numbers represent a more efficient feed conversion.9
Results shown below reflect North American feed efficiency values.
Overcoming Broiler Challenges
Additional trials explored how RFCs helped broilers improve performance in the face of health and environmental challenges, as well.
In this instance, the researchers examined the effects of clean water, mycotoxins and coccidia, as well as RFCs (50 g/MT), on broilers. The basal diet contained on average 50 ppb aflatoxin, 0.4 ppm deoxynivalenol (DON) and 2.7 ppm fumonisin. Treatments included new or used litter, clean or unchanged water and diets that did or did not include a coccidiostat.
Researchers found:
- The study demonstrated the capacity of RFCs to maintain feed efficiencies in the absence of a coccidiostat in the grower and finisher phase
- In the presence of water, litter, mycotoxin and coccidia challenges, RFCs improved the feed conversion ratio at 35 and 49 days of age.
- Under these challenge conditions, the addition of RFCs to diets in all three broiler growth phases was found to support optimal performance
Therefore, including RFCs in an animal’s diet throughout their life cycle can help improve immune function by providing a defense mechanism against pathogenic bacteria. As a result, RFCs can help maintain gut health and overall animal health, boosting immune response.
1 Food and Agriculture Organization of the United Nations. The State of Food Security and Nutrition in the World. 2017. Available at: http://www.fao.org/3/a-I7695e.pdf. Accessed February 14, 2018.
2 Hao H, Cheng G, Igbal Z, Ai X, Hussain HI, Huang L, Dai M, Wang Y, Liu, Z, Yuan Z. Benefits and risks of antimicrobial use in food-producing animals. Front Microbiol. 2014;5:288.
3 M’Sadeq SA, Wu S, Swick RA, Choct M. Towards the control of necrotic enteritis in broiler chickens with in-feed antibiotics phasing-out worldwide. Animal Nutrition., 2015;1(1). Available at: https://www.sciencedirect.com/science/article/pii/S2405654515000050.
4 Walker GK, et al. The effect of refined functional carbohydrates (RFC) from enzymatically hydrolyzed yeast on the presence of Salmonella in the ceca of broiler breeder pullets. Abstract # 17. Poult Sci 2015;94:E-Suppl.1.
5 Jalukar S, Oppy J, Davis S. Effect of enzymatically hydrolyzed yeast supplementation on performance and in protecting broilers against a mild coccidiosis challenge. Joint ASAS/ADSA meeting, 2008.
6 Brake JT. Effect of CELMANAX supplementation in diets naturally contaminated with aflatoxin and DON on broiler performance. Report on file. 2012.
7 Baines D, Erb S, Turkington K, Kuldau G, Juba J, Masson L, Mazza A, Roberts R. Mouldy feed, mycotoxins and Shiga toxin- producing Escherichia coli colonization associated with Jejunal Hemorrhage Syndrome in beef cattle. BMC Veterinary Research 2011;7:24.
8 CELMANAX study shows improved feed conversion in turkey poults exposed to production stressors. Research Notes P-82. Available at: https://ahanimalnutrition.com/-/media/spd/files/product-literature/poultry/celmanax/research/celmanax_research-notes_p-82.ashx. Accessed February 14, 2018.
9 University of Kentucky Poultry Production Manual, Chapter 6. Available at: http://www2.ca.uky.edu/poultryprofitability/Production_manual/Chapter6_Factors_affecting_feed_conversion/Chapter6_Introduction.html. Accessed February 13, 2018.



